Category A Compliant
Category B Compliant
Postal Kits
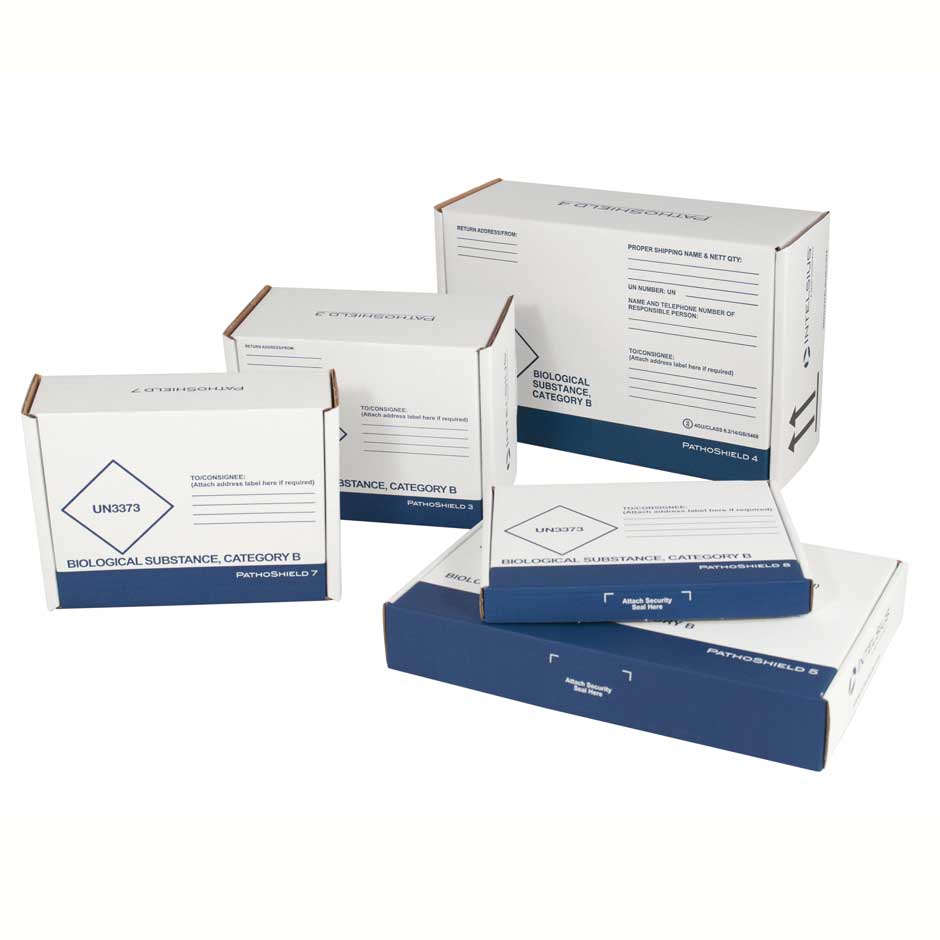
PathoShield
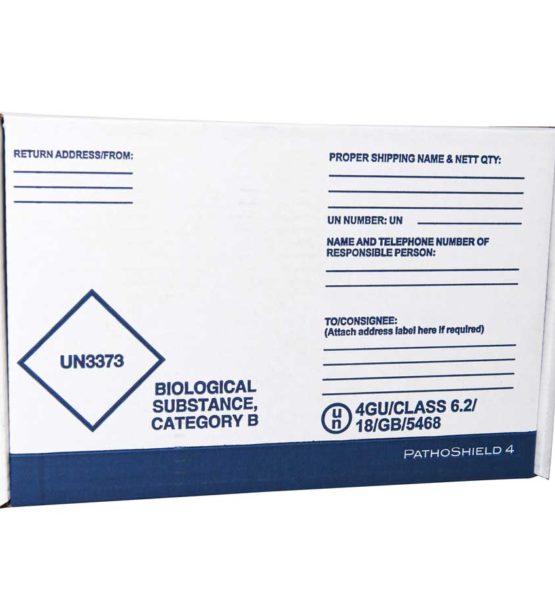
The PathoShield range are complete shipping solutions designed to carry a wide range of primaries, including blood vials, swabs (including COVID-19 swabs) and sample containers. Each system is pre-printed with the required markings and includes the relevant components to ship samples in compliance with transport regulations.
These systems are ideal for use throughout all phases of a clinical trial with ease of shipping and compliance at the forefront of design. Highly flexible in composition these systems can be suitable for transporting a wide range of samples or testing kits.
COVID-19 SOLUTIONS
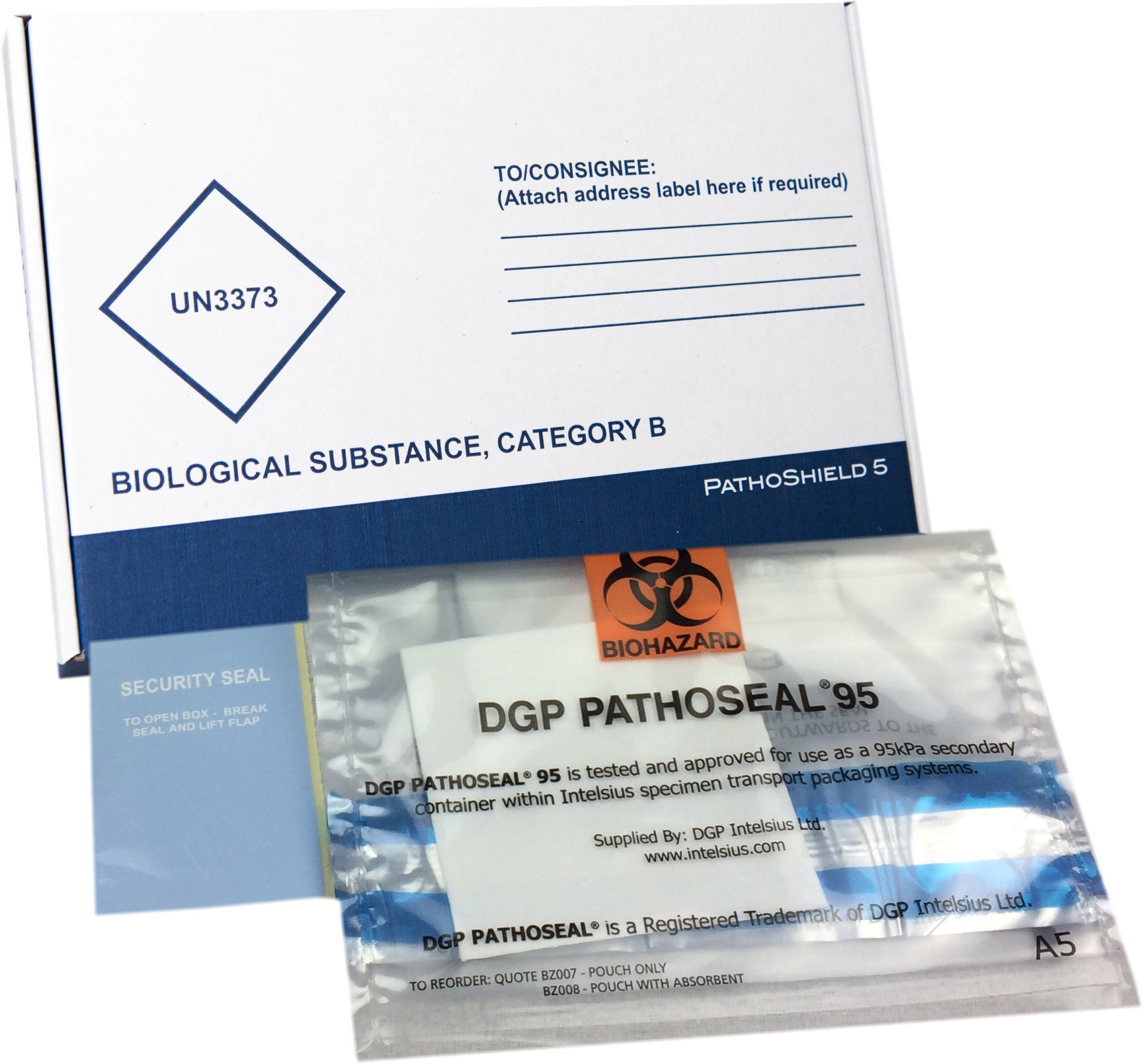
PathoShield packaging solutions are fully compliant and certified to transport Category B and Category A infectious biological materials. Samples being sent for COVID-19 testing and analysis are recommended by the World Health Organisation (WHO) to be classified as Category B UN3373 biological materials. To discuss how to transport your samples in accordance with compliance, please email compliance@intelsius.com or contact a member of our customer services team.
PathoShield Schematics
Complete Category A and Category B solutions include flexible 95kPa tested compliant secondary container, absorbent, labels, and UN markings
Range of sizes suitable for shipping through postal and courier networks
Carry a wide range of primaries including blood vials, swabs and sample containers
Can be supplied as an insulated pack
Supplied flat packed for efficient storage and shipping prior to use
Download product Sheet