The shipment of temperature-sensitive biological samples is a delicate process that requires specialist solutions. The success of clinical trials, cell and gene therapies and general patient health often depends upon the safe and secure transport of samples to and from health facilities and testing centres. Using compliant packaging solutions at the right temperature profile is essential to ensuring that your high-value samples travel safely and securely along the cold chain.
This article addresses five common pain points when shipping temperature-sensitive samples and how to overcome them.
Maintaining temperature
If you’re conducting clinical trials, shipping human samples for testing, or developing cell and gene therapies, it’s vitally important to transport samples within the specified temperature ranges to avoid any damage or loss of efficacy. Passive temperature control is typically used to transport temperature-sensitive samples to and from test sites, labs and healthcare settings.
Properly deployed, temperature-controlled packaging, combined with specialist logistics services, can be the difference between positive and negative patient outcomes or the success or failure of clinical trials. Passive temperature-controlled packaging typically consists of an outer correx or cardboard box, insulation such as expanded polystyrene (EPS), or vacuum insulated panels (VIP) which prevents external temperatures from affecting the chosen internal temperature, and a water-based, plantol-based or dry ice phase change material that sets the internal temperature range.
What are PCM, EPS, and VIP, and why are they used in passive packaging?
PCM
PCM is used in passive packaging to stabilise temperature, either to keep the payload from warming up too fast, or to keep it from getting cool too quickly. You can think of PCMs in passive packaging in the same way you would a box cooler you would use during a picnic on a hot day. When you place food or cold drinks into a cooler with ice packs, it helps to keep the temperature in the cooler relatively stable, until the ice in the packs has fully melted.
The temperature of the items in the cooler will only start to increase once all the excess heat has been absorbed by the cool packs. PCMs work in a similar way in passive packaging. The fully frozen PCM will absorb excess thermal energy (heat) within the packaging, which helps to keep your precious samples at a stable temperature throughout transit.
If you would like to understand further, click here to access our white paper on how PCM works.
EPS
EPS is a rigid, foam material made from solid beads of polystyrene and is made up almost entirely (98%) of air. The trapped air is a very poor heat conductor, making EPS an excellent insulator. It is also extremely lightweight, making it a very viable option for shipping. The Intelsius BioTherm and PharmaTherm solution ranges utilise EPS as insulation.
VIP
VIPs are used in a variety of industries from construction to refrigeration, as a form of thermal insulation. VIPs comprise a gas-tight enclosure which acts as a vacuum to prevent air or water vapour from entering it. It is usually made up of very porous material like fumed silica or aerogel and has thin walls which makes it lightweight and effective for use in long-haul shipping.
An example of a combination of PCM and VIP is the ORCA range: a high-performance packaging system that utilises advanced PCM and VIP, providing better temperature protection than when compared with water-based systems. The range covers temperature ranges from -70°C with dry ice to 15-25°C and is available in an array of sizes for all your payload needs.
The combination of PCMs and VIPs like those used in ORCA solutions mean that for particularly sensitive or high-value sample shipments, you can be assured of only the highest-performance of temperature control for a longer duration, without the need for human intervention during transit.
The labelled diagrams below illustrate where a payload is placed within an ORCA Multi-Use and Single Use packaging system respectively:
ORCA Multi-Use
ORCA Single-Use
| Features of ORCA Multi-Use | Features of ORCA Single Use |
|---|---|
| Qualified and tested against challenging ISTA 7D profiles for a minimum of 96 hours | Qualified and tested against challenging ISTA 7D profiles for a minimum of 96 hours and a minimum of 120 hours when using dry ice as a coolant |
| Utilises advanced PCM and VIPs to avoid temperature excursions | Utilises advanced PCM and VIPs to avoid temperature excursions |
| Convenient, fast and identical pack out processes for both summer and winter seasons | Universal pack-outs |
| Strong and robust outer with compact internals to reduce weight | Strong and robust outer with compact internals to reduce weight |
| Up to a 5-year life cycle for reuse and refurbishment | One-time use |
| Available in a range of sizes and temperatures to suit all payloads | Available in a range of sizes and temperatures to suit all payloads |
Compliance
Adhering to international standards on the transport of temperature-sensitive goods is of paramount importance to avoid legal and regulatory consequences. As the shipment of biological samples affects human life and health, compliance with international regulations like those imposed by The International Air Transport Association (IATA) and The European Agreement concerning the International Carriage of Dangerous Goods by Road (ADR) is essential to prevent compromising the integrity of high-value samples and to ensuring that your shipment does not fall short of those standards.
The United Nations has issued guidelines for the shipping of infectious substances, as follows:
Category A and Category B (UN3373)
The UN guidelines split the shipping of infectious substances into two separate categories: Category A and Category B
Category A is defined as an infectious substance that, when exposure to it occurs, has the potential to cause permanent disability or a life-threatening or fatal disease in humans or animals. Category A is further divided into two sub-categories:
UN2814 (affecting humans) and UN2900 (affecting animals only)
UN2900 (affecting animals only)
Category B (UN3373) is described as any substance which does not fall into Category A. When referring to compliance, we are referring to the labelling and packaging compliance required to ship Category B (UN3373) samples.
We have created a helpful UN Class 6.2 Classification which you can access here if you are unsure which Category your sample falls into.
You can also access the complete guide on how to ship UN3373 samples by clicking here.
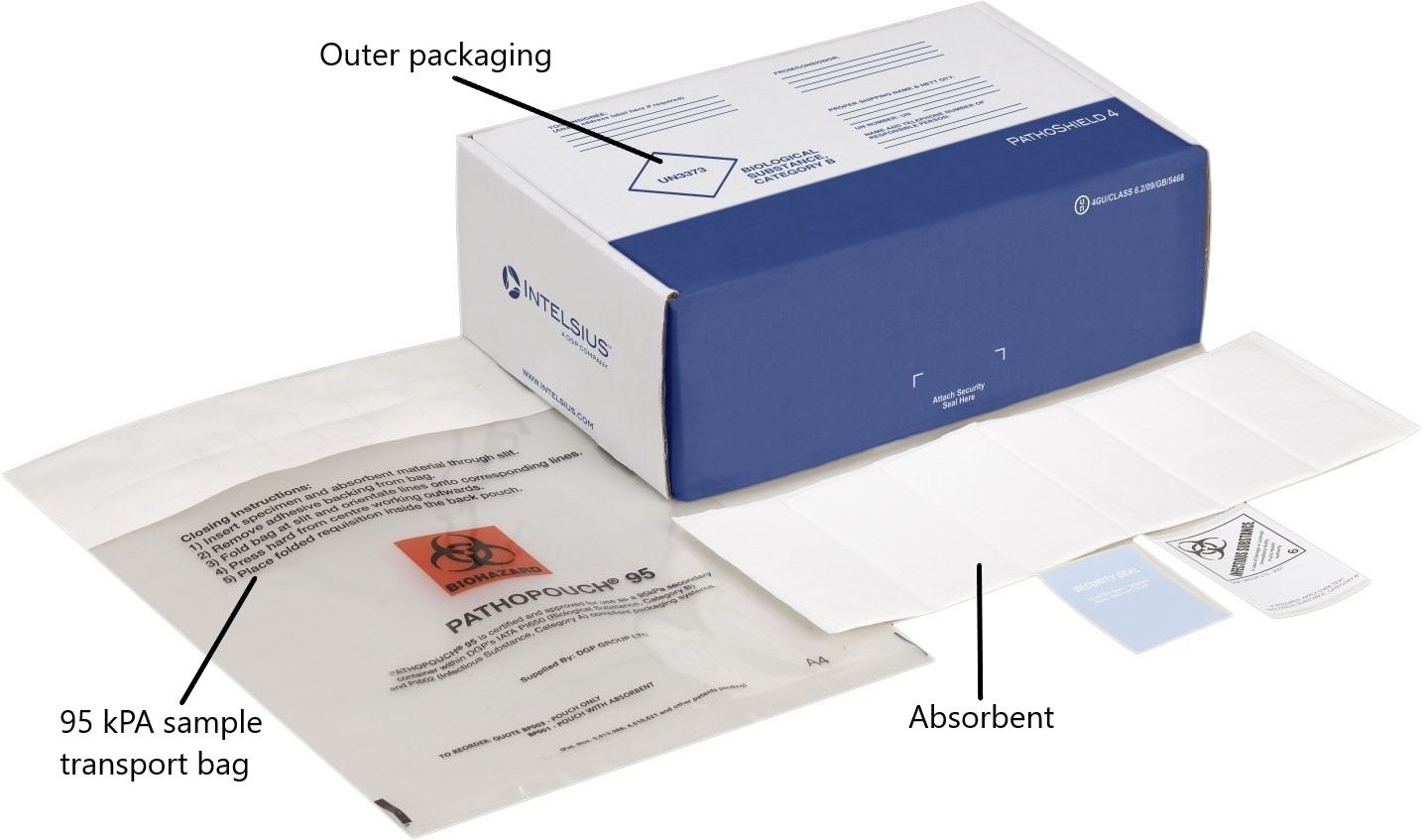
Protecting samples
As samples are being transported to different destinations, they will traverse a range of terrains and may be subject to bumps or shock along the way, resulting in damage to the payload or even leakage of samples. The type of protective bag used is key to prevent leakage. Intelsius uses sample bags that are in accordance with IATA guidelines on UN3373 sample shipping, which states that they must be able to withstand 95kPa of pressure. These sample bags are used to secure primary containers when the payload contains liquid, in order to prevent leakage. Additionally, the outer materials used in the packaging should be durable, sturdy and able to withstand shocks.
Besides adhering to the labelling requirements above, shipments must adhere to packaging guidelines known as Packing Instruction 650 (P650 or PI650), which is divided into guidelines for ADR and IATA and set out a list of requirements covering the quality and construction of the packaging used for transport of UN3373 samples.
The guidelines stipulate that the packaging must comprise three components:
- A leak-proof primary receptacle(s) containing not more than 1L / 1kg (multiple primary receptacles must be individually wrapped or separated)
- Leak-proof secondary packaging
- A rigid outer packaging with one surface having minimum dimensions of 100 x 100 mm.
The packaging must be clearly labelled with the UN3373 diamond, while the words “Biological Substance, Category B” written adjacent to it. When transporting liquids, sufficient absorbent must be placed between the primary and secondary packaging to absorb all the liquid content in the primary receptacle. A maximum of 4L per package is allowed. For solids, both primary and secondary packaging must be sift proof, and a total of 4kg per package is the maximum allowed.
Below are two examples from Intelsius’ sample transport line of products which are Category A and Category B compliant:
PathoShield
The PathoShield range is designed to ship a wide range of primaries including blood vials, swabs, and sample containers. Each system is pre-printed with all the required markings and includes all necessary components to compliantly ship your samples. Pathoshield systems are both Category A and B compliant and can be used alongside the 95kPa Pathopouch, and Pathoseal 95 ranges to ship UN3373 Category B samples.
PathoPak
The PathoPak range is UN-certified to ship Category A and B samples and meets all necessary international transport regulations, including IATA, ADR and CFR 49 (DOT). The systems are available in a range of sizes from 800ml to 3L and its full schematics can be viewed here.
Tracking shipments/samples
Aside from ensuring that the packaging used to ship samples adheres to international safety standards, it is equally crucial to be able to monitor the movement of your payload as it makes its way to its final destination. Key data such as temperature, humidity, shock, and light are essential to not only give you peace of mind about the status of your samples but also intervene and rectify any temperature excursions that occur during transit.
It is best to select a packaging solution equipped with a reliable data logger which allows you to track and monitor essential data. This data should also be available in real-time and should be easily accessible, such as with ORCA Connect. With a designated space for a data logger, you can access key information as the shipment moves across its journey.
To make accessing your payload data user-friendly and convenient, we have created ORCA Cloud, a universal data logger portal that allows you to record all your logger devices in one place. Regardless of the number of loggers used, you can access all the data however and whenever you need to. ORCA Cloud is compatible with all logger types and the data is presented in one, consistent manner for your reference. To find out more, click here.
| Key Features of ORCA Connect |
|---|
| Tested against ISTA 7D temperature profiles |
| Qualified for up to 96 hours |
| Dedicated space to place data logger |
| Available in a range of sizes across multiple temperature profiles |
| Available to purchase or rent |
| Potential real-time tracking facilities for: External and internal temperature Exact location Orientation Battery status |
Documentation and labelling
Is a Shipping Declaration required for Category A and B shipments?
P650 Instructions state that you do not need a Shipper’s Declaration for Category A and B shipments, however you would require other basic documentation, depending on which method of shipment your samples are being sent through.
For example, if transporting by air, an Airway Bill is required and it must indicate the name and contact number of the person responsible for the packaging, and the name and address of the shipper and consignee must be on each package.
Intelsius designs complete transport sample solutions, with all necessary and correct labelling included in our service, however we have created a helpful label guide for your reference should you need assistance with label compliance.
Get in Touch
To find out more about our full range of temperature-controlled and sample transport packaging solutions, visit our website here or reach out to our friendly customer service representatives at cs@intelsius.com who can assist you with any queries.