In the first of three blog posts exploring biopharmaceutical trends to look out for in 2021, we’ll delve into digital transformation and artificial intelligence (A.I.) in the biopharmaceutical research and development (R&D) industry, including Bioprocessing 4.0, and how it’s being deployed to bring biopharma into the 21st century.
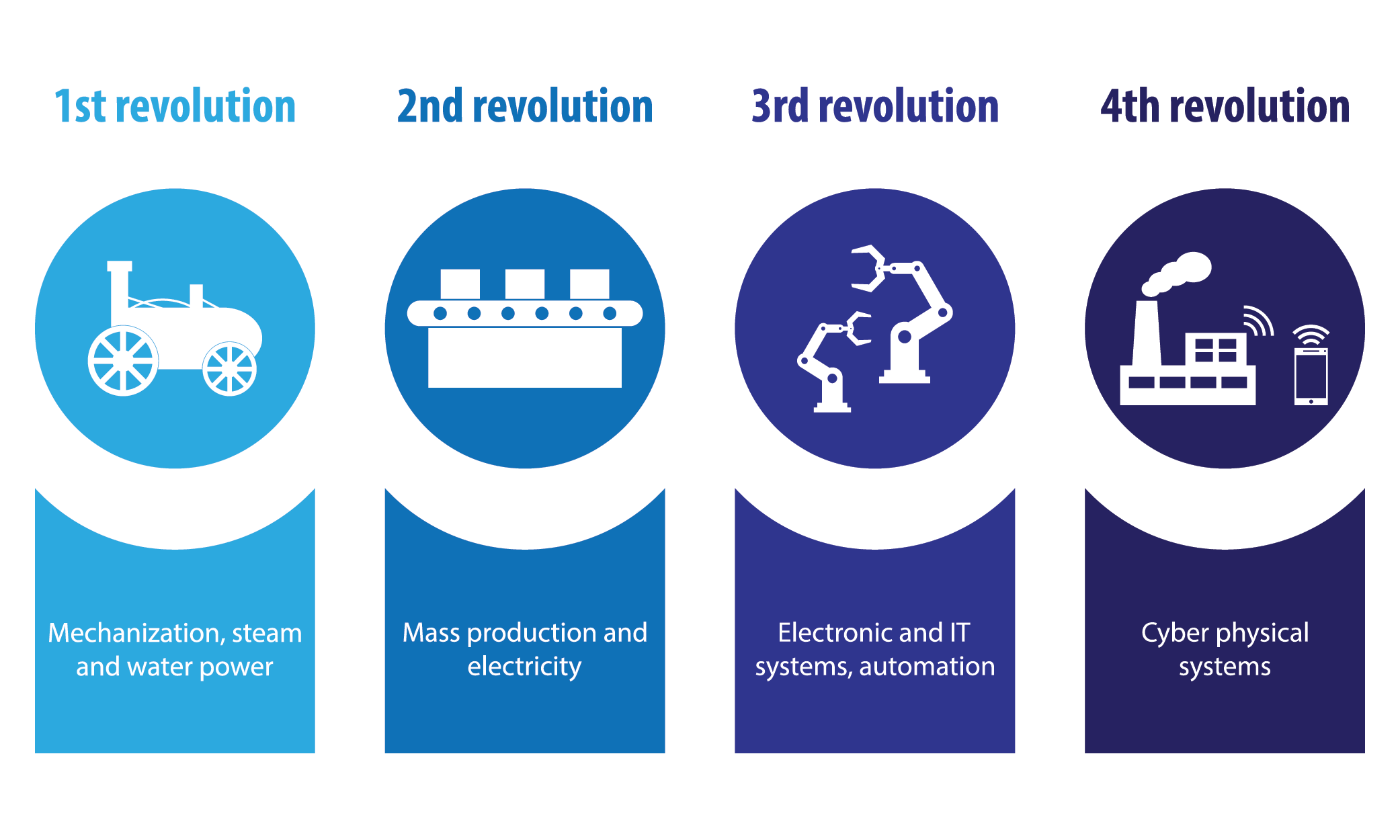
Industry 4.0: The Fourth Industrial Revolution
First came steam and water power, then electricity and assembly lines, and finally computerisation arrived to give us the third industrial revolution. With the arrival of robotics, A.I. and smart technology, we’re ready to embrace the arrival of the fourth industrial revolution, often referred to as Industry 4.0.
This latest technological advancement has equal if not more potential to alter the way we live than the previous industrial revolutions. Put simply, Industry 4.0 focuses on connectivity and automation, with internet connected machines interacting, learning from each other, and making decisions autonomously. You could be forgiven for confusing Industry 4.0 for a sci-fi plot line, but this latest industrial sea-change is already beginning to influence the way we live in a real way.
Smart homes are able to read temperature changes and adjust heat and cold accordingly. Your phone, laptop and TV can be connected, and managed from clouds or similar internet-based device management systems that learn from your choices and make recommendations based on their ongoing learning. Fridges, washing machines, car keys and even children’s toys are being fitted with A.I. For the first time, young people are growing up with the expectation that everything connects to everything else – the days of adapters and analogue fixes seem to be coming to end.
What it Means for the Biopharmaceutical Industry
There are good reasons for the biopharmaceutical industry lagging behind other sectors when it comes to integrating A.I. and general digitisation within the fabric of its operations. Unlike the finance, oil, and gas sectors (who have all invested heavily in A.I. over the past two decades), the research and development of complex biopharmaceutical products for human use is heavily regulated, with extensive safety protocols attached to each and every process. With myriad variables involved in the testing and development process behind drug development, integrating non-human processes at the scale and with the diligence required to develop pharmaceutical products safely and effectively is incredibly complicated.
That said, A.I. is starting to find its feet in biopharmaceutical R&D. While the push towards automation and digitisation pre-dates COVID-19, the desire to bring about an end to the SARS-COV-2 pandemic has intensified the drive towards automation. This new need for speed without compromising safety and quality has forced R&D companies to invest further than ever before into technologies that could help streamline their processes.
But COVID-19 isn’t the only diver pushing the biopharmaceutical industry towards digitising and automating its processes. Costs, carbon footprint, manpower, time, and risk can all be reduced with this smart technology, as when properly implemented, Industry 4.0 can significantly impact the way you work, providing intelligent feedback that allows you to cut unnecessary steps from your production chain, and streamline others.
One of the better-known expressions of biopharmaceutical automation is something called Bioprocessing 4.0, which focuses on how to apply today’s data, analytics, artificial intelligence, automation, and technology for the digital transformation of biologics manufacturing.
Bioprocessing 4.0.
As with most industries, biopharma is always on the lookout for ways to do things faster, more cost effectively, and more safely – or at least go faster and at less cost without compromising existing safety standards. This idea isn’t anything new. In fact, the concept of process intensification has influenced manufacturing techniques for over 100 years, and in basic terms involves the analysis of your manufacturing processes with a view to finding new, time and cost-efficient solutions to optimise your manufacturing.
You may have seen this occasionally referred to as ‘lean kaizen’, a process developed by Japanese car manufacturer, Toyota after World War 2 which ultimately led to significant improvements to Toyota’s manufacturing, saving them time and money. However, coupling process intensification with digital transformation is a relatively new concept to biopharma. Bioprocessing 4.0 takes these principals and combines them with cutting edge technology to create truly connected and intelligent automation within existing manufacturing processes to speed up and improve biopharma research and development.
Derived directly from Industry 4.0, it wasn’t until 2018 when the term ‘Bioprocessing 4.0’ began being talked about in biopharmaceutical circles, as an end-to-end, connected biopharma process designed to improve all aspects of the R&D process. Crucial to making Bioprocessing 4.0 a reality is digitising previously analogue processes, which in this context is known as a ‘digital twin’.
Digital Twins
A digital twin is commonly understood as a virtual representation of a real-world process that allows understanding, optimization and monitoring of the process.1 The creation of this digital twin is a core part of Bioprocessing 4.0, as it allows users to pull live data from the entire R&D process, making changes quickly and efficiently, solving problems before and as they arise, and removing unnecessary steps that slow you down.
In practice, this means highly sensitive sensors feeding live environmental data into a cloud or main computation platform, which can read and understand this data without human intervention. But the next phase of this digital loop is the really smart part: upon receiving this constant stream of live data, a fully integrated Bioprocessing 4.0 system can then use this information to send instructions directly to the instruments, streamlining processes, fixing problems and ensuring no time is wasted when potential issues occur.
Without this digitised approach, spotting and fixing issues throughout the various processes involved in biopharma R&D can involve long periods of shut down, while skilled engineers diagnose the problem, before manually correcting it. Beyond digitisation, though, Bioprocessing 4.0 has another key feature: internet connectivity. Within the Bioprocessing 4.0, all the machines throughout your manufacturing process are connected together wirelessly, which are in turn wirelessly connected to a main computer. This level of connectivity is known as The Internet of Thins (IoT).
The Internet of Things (IoT) in a biopharmaceutical context is best described as having all your manufacturing elements connected via the internet. In terms of Bioprocessing 4.0, that means not only are you pulling live data and simultaneously feeding it back into manufacturing processes, but you’re doing so in a completely connected way. Everything in the R&D process is connected and allows for your digital twin to have full oversight of your various processes, not in silo, but in unison.
Sanofi: A Case Study in Bioprocessing 4.0
One example of how Bioprocessing 4.0 is impacting the biopharmaceutical industry is Sanofi’s Massachusetts based biomanufacturing facility opened in 2019, described as a ‘one of the world’s first’ to implement this technology. Franqui Jimenez, PhD, head of second-generation process development, global manufacturing science and technology at Sanofi describes how the facility has already had big benefits for Sanofi: ‘The advanced, paperless, and data-driven manufacturing technologies enable achievement of higher levels of productivity, agility, and flexibility, which reduces the time needed for products to get from the development labs to the manufacturing plant.’2
Described by Sanofi as a ‘factory of the future’, they have also reported up to an 80% reduction in energy usage and carbon dioxide emissions, and reductions of up to 90% or more of water and chemical usage as a result of implementing Bioprocessing 4.0 in this facility.3
‘The Framingham facility is a lighthouse in Sanofi’s strategic transformation of its global manufacturing platform,” said Brendan O’Callaghan, Global Head of Biologics at Sanofi. ‘This facility allows us to connect great science, differentiated medicines and the best in cutting edge, digitally-enabled manufacturing technologies.’4
In Summary
Clearly, Industry 4.0 has well and truly arrived, and so it was only ever a matter of time before the biopharmaceutical industry caught up. With the introduction of Bioprocessing 4.0, it seems as though biopharmaceutical companies will finally join the fourth industrial revolution. Certainly, with costs and environmental to be reduced, and for high-quality biopharmaceutical products to be made at the speed, volume, and efficacy required to satisfy growing markets, we can expect to see more factories of the future popping up around the world.
In our next blog looking at biopharma trends to loom out for in 2021, we’ll be exploring the role of biosimilars, and how they’re changing the landscape of the biopharmaceutical industry.
To ensure you don’t miss any of our future articles, follow our LinkedIn and Twitter social channels for regular updates.
External References
- Go Silico (2020) – Digital Transformation and BioPharma 4.0
- Gen News (2020) – Coordinating the Move to BioProcessing 4.0
- Sanofi – (2019) – Factory of the Future
- Sanofi (2019) – Factory of the Future